Cologenesis Colo Gide GTR Membrane Pack of 1 Pc
CologGide GTR Membrane (Sterile Collagen Sheet)For guided Tissue Regeneration of bone damageCologenesis Colo Gide GTR Membrane Pack of 1 Pc #10x15 mm
| Expiry Date | Feb, 2027 |
Cologenesis Colo Gide GTR Membrane Pack of 1 Pc #15x20 mm
| Expiry Date | Oct, 2026 |
- Osteostimulation & Osteoconduction
- 100% Synthetic Antimicrobial (transient, local)
- Anti-Inflammatory (transient, local)
- Room Temperature Storage
The ColoGide is a resorbable, non-friable barrier membrane obtained from highly purified type-I collagen derived from specially controlled and certified animals and is highly purified to avoid any antigenicity.
The cologide is produced to provide an optimized degree of flexibility and rigidity combined with the most predictable resorptive process.
The product comprises two distinct surface textures on either side of the membrane-a dense surface and a roughened porous surface. The dense top layer is in contact With soft tissue to prevent epithelial down growth and the porous roughened surface is in contact with bone and favors bone ingrowth.
Indications:
- Periodontology- guided tissue regeneration procedure for infrabony defects.
- Oral implantology-guided bone regeneration procedures for sinus graft procedures, ridge augmentation, and peri-implant defects.
- Augmentation procedures- horizontal and vertical ridge augmentation or augmentation of the maxillary sinus.
- Surgical root resection, cystectomy and other bone lesion.
Contra-Indications: The cologne should not be used in all contraindications customary to the use of bone grafts that can impair bone or wound healing including acute or chronic bone infection, uncontrolled diabetes, chronic high dose steroid therapy, vascular impairment at the implant site, metabolic bone disease, and alcohol are smoking abuse.
Storage: It should be stored in the original packaging in a cool dry place at room temperature.
Available Size
- Sizes: 10X15mm
- Sizes: 15X20mm
- Sizes: 20X32mm
- Sizes: 30X42mm
Cologenesis Colo Gide GTR Membrane Pack of 1 Pc
- 1 x Sizes 15X20 mm
In all cases standard established sterile surgical procedures must be. A full-thickness mucoperiosteal flap must be raised to explode the underlying bone defect. The defect must be debrided thoroughly of all granulation tissue. After adequate site preparation, the bone defect can be filled with graft material. The membrane once wet with blood tends to adapt extend 2-3mm beyond the edge of the defect. The membrane should be properly immobilized as micro-movement can impair bone ingrowth. immobilization can be done using suturing or a fixation system. The wound should be primarily closed o0ver without any lesion in the flap.
Related
Products
Mani Diamond Burs - Ball Round…
Diamond-coated dental burs for precise cavity…
Neoendo Flex Coronal Flaring…
25mm Niti Heat treated highly flexible neoflex…
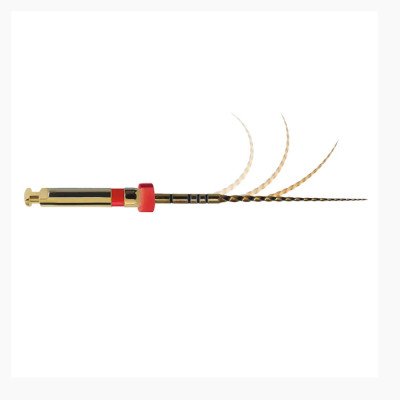
Neoendo Flex Files 25mm 6% #40…
25mm Niti Heat treated highly flexible neoflex…

Mani Diamond Burs - SI - 47C…
Diamond-coated dental burs for precise cavity…

Neoendo Flex Glide File 21mm 2%…
2% Taper Niti Heat Treated Highly Flexible…
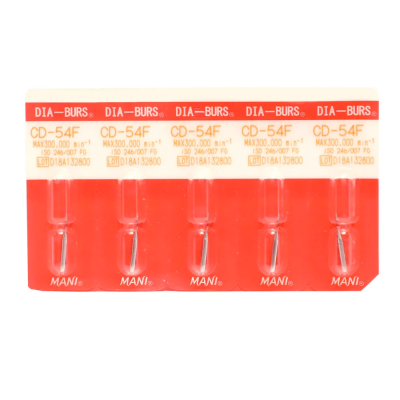
Mani Diamond Burs - Children'S…
Diamond Burs - Children'S Dentistry - Cd Series…

Mani Diamond Burs - TR - 62C…
Diamond-coated tapered round dental burs for…
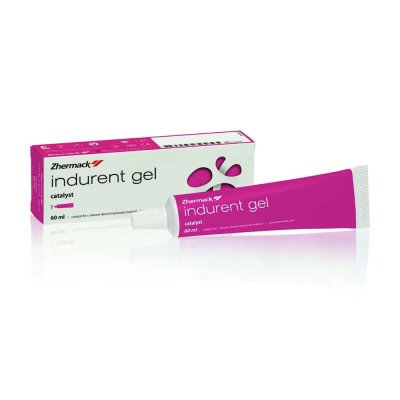
Zhermack Zetaplus Indurent…
Catalyst gel for C-silicone impression material…
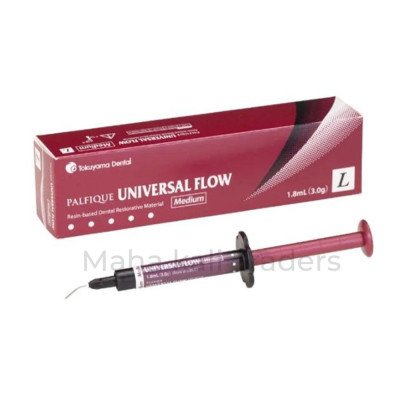
Tokuyama Palfique Universal…
Effortless application, rapid curing, and…